Welcome back to Pulm PEEPs Fellows’ Case Files series. We are traveling to the midwest to visit The Ohio State University College of Medicine and hear about another great pulmonary case.

Meet Our Guests
Kashi Goyal is a second-year Pulmonary and Critical Care Fellow at The Ohio State University Wexner Medical Center. She obtained her MD at OSU, and then completed her Internal Medicine residency at Beth Israel Deaconess Medical Center. She worked as a hospitalist and educator before going back to fellowship and remains passionate about medical education.
Lynn Fussner is an Associate Professor of Internal Medicine at OSU and has been there since completing her fellowship and Post-doctorate at Mayo Clinic. In addition to her clinical work in the multidisciplinary vasculitis clinic, she is a translational researcher with a focus on inflammatory pulmonary disorders and vasculitis.
Avi Cooper is an Assistant Professor of Medicine at Ohio State University College of Medicine and the Program Director of the Pulmonary and Critical Care Fellowship. He is an Associate Editor at the Journal of Graduate Medical Education. Last but not least, he co-hosts the Curious Clinician Podcast, one of the most popular medical education podcasts.
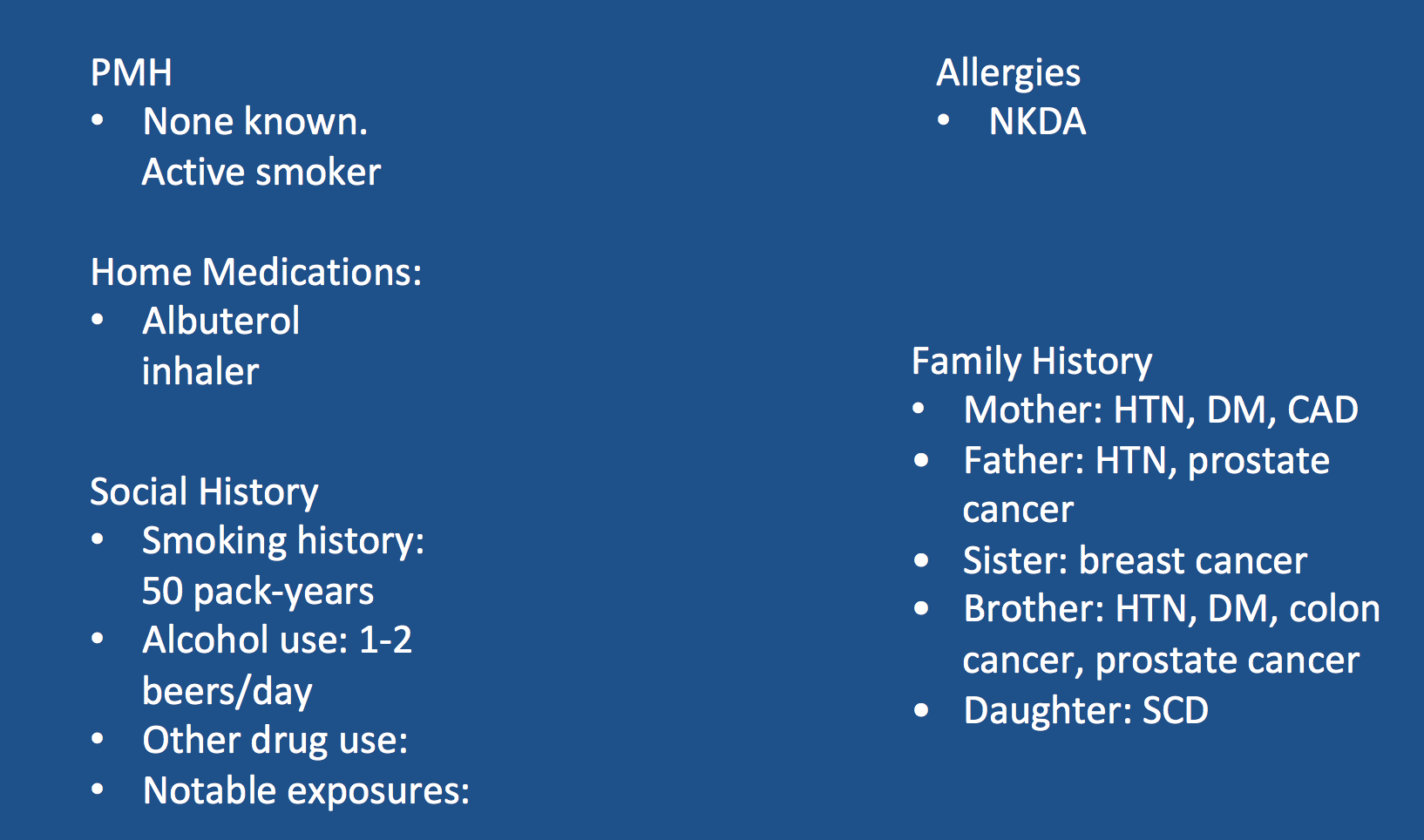
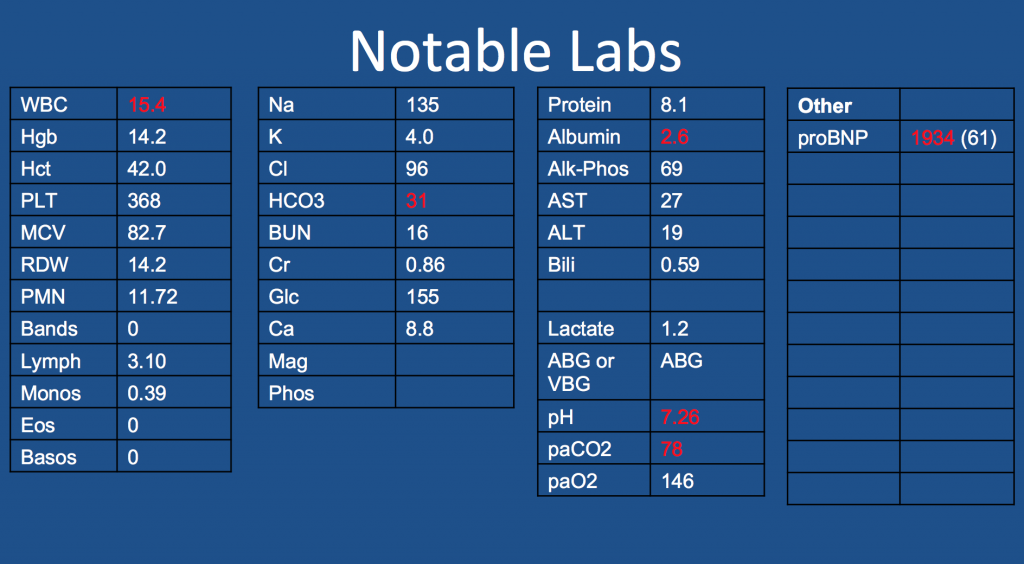
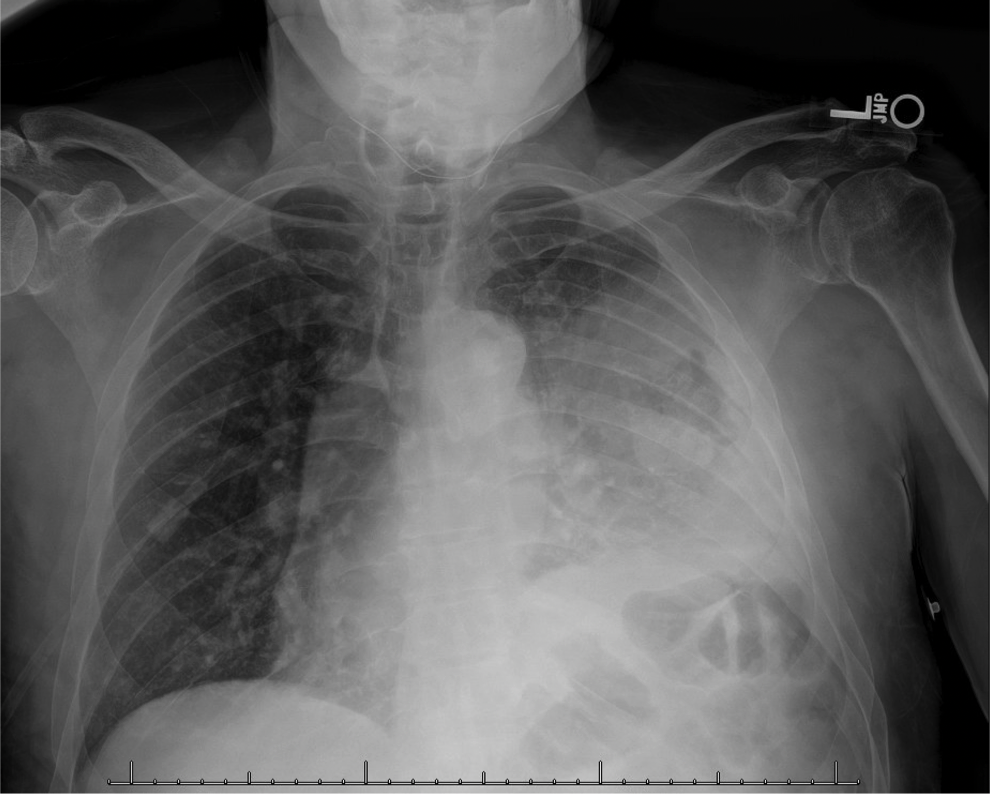
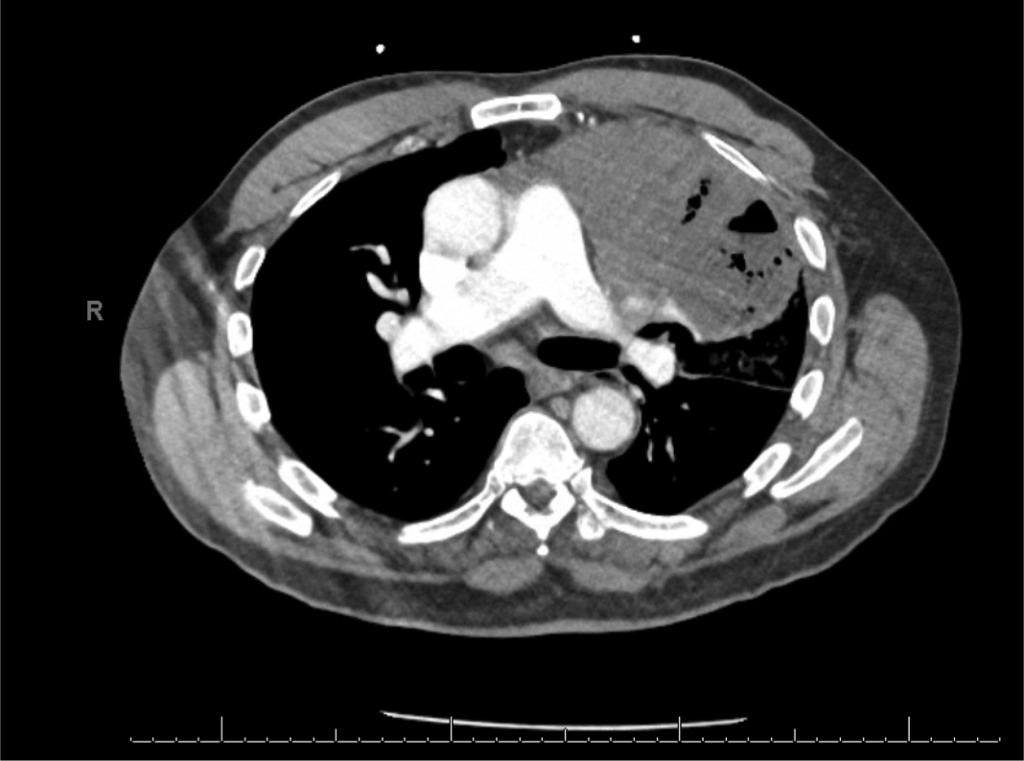
Patient Presentation




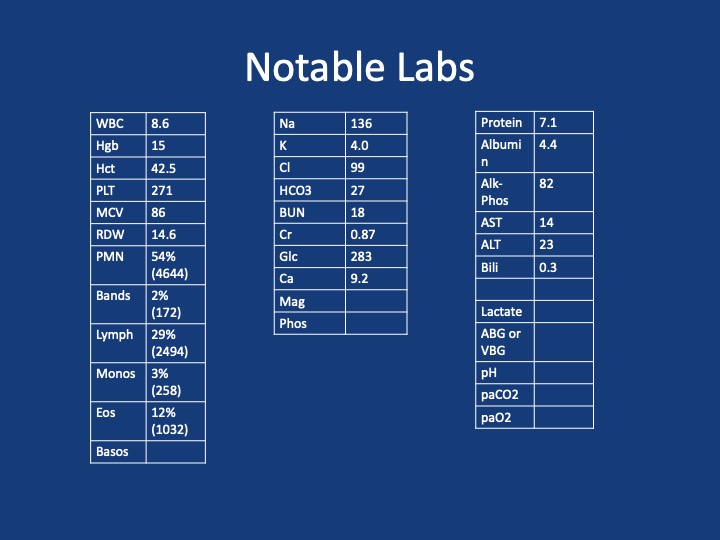
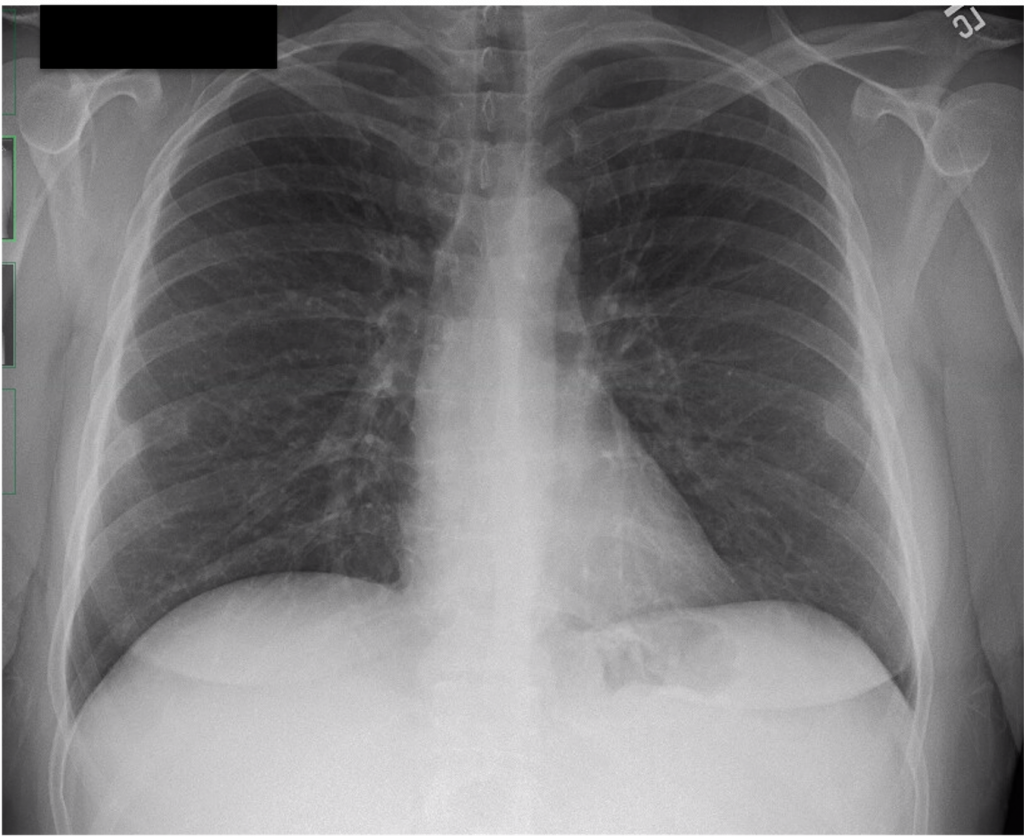
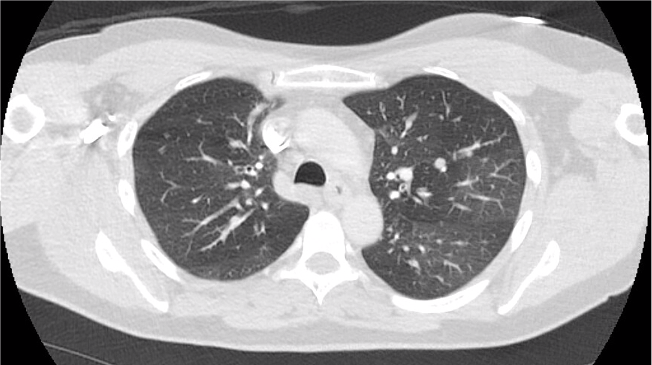
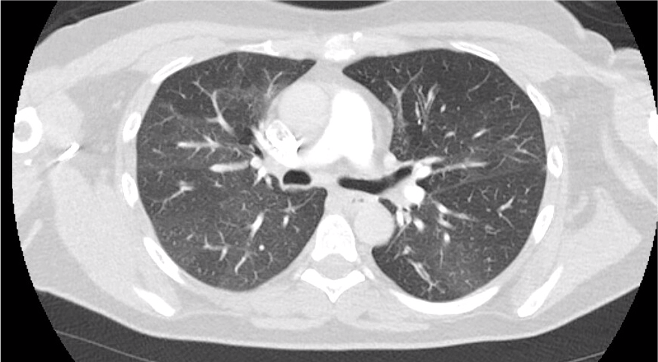
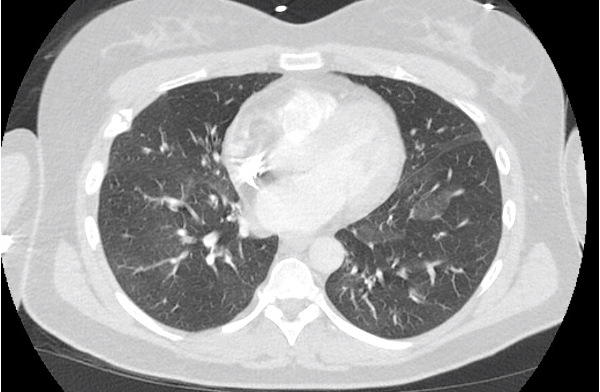
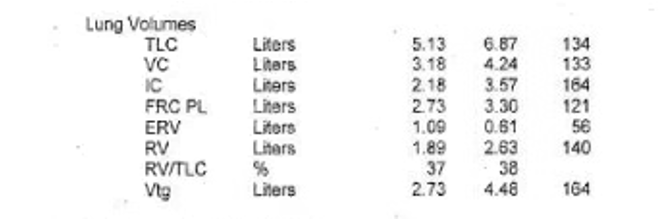
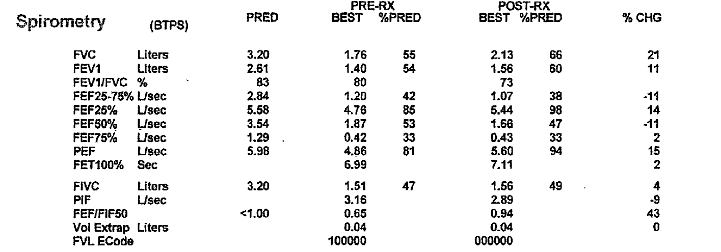
Key Learning Points
**Spoilers Ahead** If you want to think through the case on your own we advise listening to the episode first before looking at these points.
- The three most common causes of cough in adults in the USA are cough variant asthma, GERD, and post-nasal drip
- A post-viral cough can last for 8-12 weeks and still be within normal
- Sinus symptoms in a chronic cough can just be sinusitis and post-nasal drip, but should consider eosinophilic granulomatosis with polyangiitis (EGPA), aspirin exacerbated respiratory disease (AERD), cystic fibrosis, or ciliary dyskinesia.
- Examination of a wheeze
- Fixed sound vs variable
- Pitch: larger central airways vs lower peripheral airways
- Is it throughout the cycle or at a certain phase?
- Ask the patient to cough before listening and ask them to breathe out through their mouth
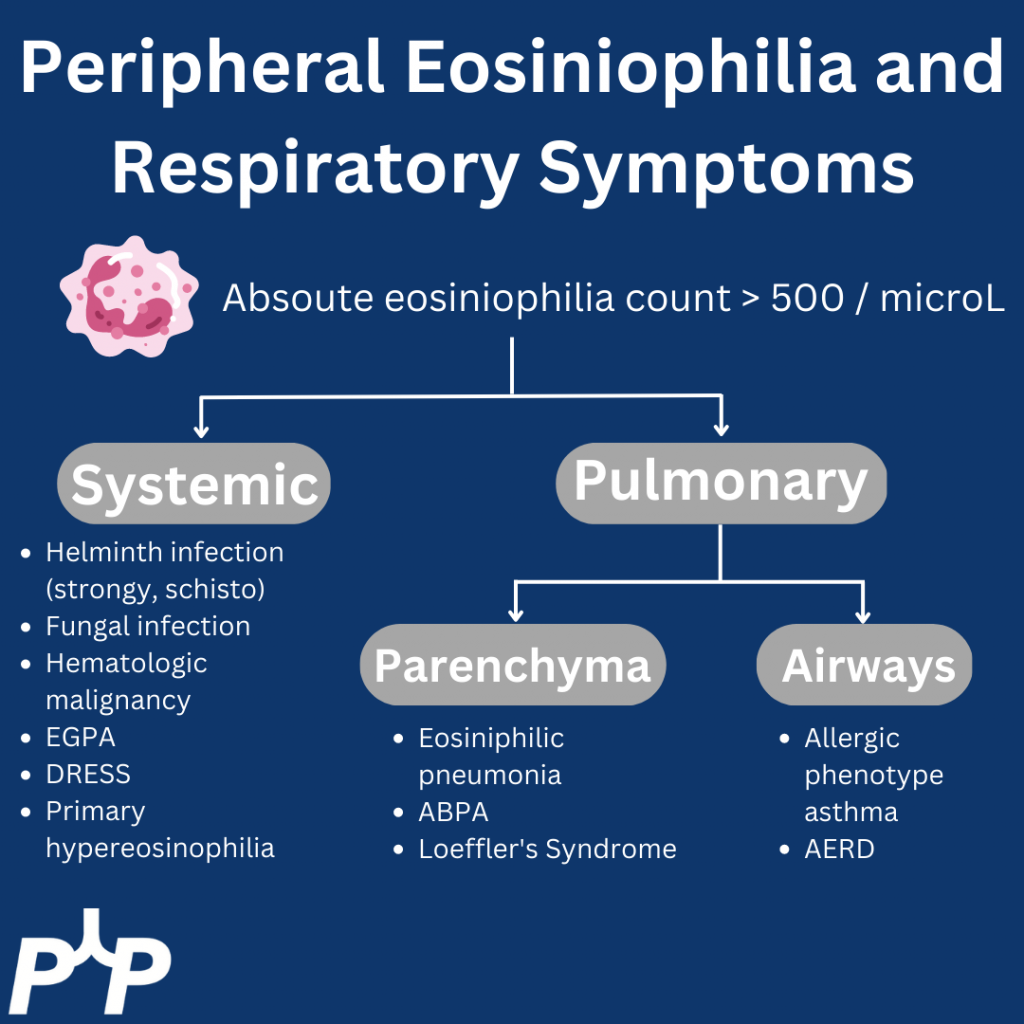
- Approach to eosinophilia in a patient with cough and dyspnea
- Multi-system involvement vs lungs
- Multi-system involvement
- Vasculitis
- Parasitic infection
- Hematologic malignancy
- Medication side effect
- Primary hypereosinophilic syndromes
- Within the lungs:
- Parenchymal disease
- Loeffler’s syndrome
- Eosinophilic pneumonia
- Airway disease
- Asthma
- ABPA
- Parenchymal disease
- Multi-system involvement
- Multi-system involvement vs lungs
- If you have a high suspicion for airways disease, PFTs should be requested with bronchodilator testing regardless of the degree of obstruction on baseline spirometry
- Asthma alone should not cause ground glass opacities, so if see these in a patient with asthma we think about:
- Infection, especially atypical infections
- EGPA
- Vasculitis with DAH
- ABPA
- Hypogammaglobulinemia or other immunodeficiency
- EGPA diagnosis
- ANCA testing is only positive in 60% of patients with EGPA so a negative test doesn’t rule it out by any means
- It is easiest to make a diagnosis when there is a clear small vessel manifestation
- Alveolar hemorrhage
- Mononeuritis multiplex
- Glomerulonephritis
- Many patients with asthma, nasal polyposis, and high peripheral eosinophilia have EGPA but don’t have a clear small vessel feature of vasculitis or a positive ANCA
- These patients typically have eosiniophilia a lot higher than when thinking about allergic phenotype asthma alone. As a rule of thumb, at least an absolute eosinophil count > 1000
References and Further Reading
- Carr TF, Zeki AA, Kraft M. Eosinophilic and Noneosinophilic Asthma. Am J Respir Crit Care Med. 2018;197(1):22-37. doi:10.1164/rccm.201611-2232PP
- Cottin V. Eosinophilic Lung Diseases. Clin Chest Med. 2016;37(3):535-556. doi:10.1016/j.ccm.2016.04.015
- Grayson PC, Ponte C, Suppiah R, et al. 2022 American College of Rheumatology/European Alliance of Associations for Rheumatology Classification Criteria for Eosinophilic Granulomatosis with Polyangiitis. Ann Rheum Dis. 2022;81(3):309-314. doi:10.1136/annrheumdis-2021-221794
- Wechsler ME, Akuthota P, Jayne D, et al. Mepolizumab or Placebo for Eosinophilic Granulomatosis with Polyangiitis. New England Journal of Medicine. 2017;376(20):1921-1932. doi:10.1056/NEJMoa1702079
Podcast: Play in new window | Download
Subscribe: Apple Podcasts | Spotify | Amazon Music | Android | iHeartRadio | Podcast Index | RSS | More